 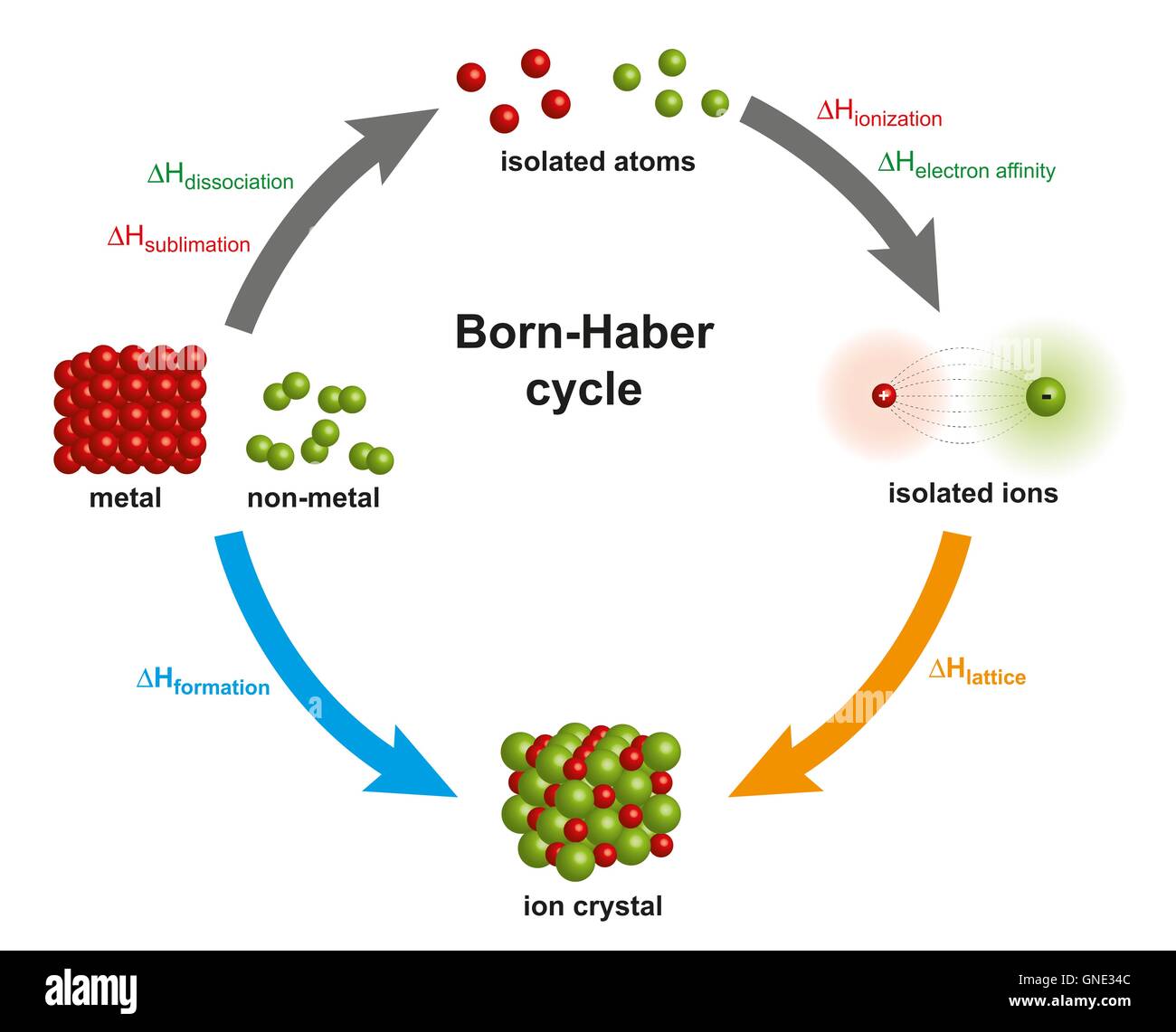
And if we reverse this situation and we have MG to positive plus two electrons gives us MG. So the delta X value corresponds to this is equal to minus +2337 minus +23 3 7 kg. So the third equation will be formed similarly, the if we put that is MG we I haven't just converted into MG two positive plus two electrons. That is if we reverse this equation to and again multiplied by two. And actually it is the reverse equation of two multiplied by two. So this value corresponds to -100 sorry, -480 kg of basically this is the third equation. 
It is a measure of the cohesive forces that bind ionic solids. So this is the second equation now to L two cl negative is converted into two cl negative is converted into cl two plus two electrons. In chemistry, the lattice energy is the energy change upon formation of one mole of a crystalline ionic compound from its constituent ions, which are assumed to initially be in the gaseous state. So this is the equation one equation, one, so now half one by two cr 21 by two Cr two plus electron gives us gives us cl negative ions and delta X value for this is equal to 2 40 kg to 40 kg. So delta X is equal to minus 643 -643 kg. So MG that is magnesium Plus CL two Cierto gives us MGs here to give us mg cr two. So the delta X value that we have to calculate that we have to calculate. So we know that MG two positive mg to positive blurs to cl negative gives us mg cr two MG cr two. sarthakparida147 Read Discuss Lattice Energy is the energy required to break apart an ionic compound into its gaseous ions. So yeah, starting from its relations with the basically latest energy of MG cR two learn this energy empty cl clue. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
